Part of Thermo Fisher Scientific
Material Safety Data Sheet
Optional Products
Organisms
Organisms in the Clinical sector this product works with:
For this Organism
Other products used in the isolation of Escherichia coli O157:
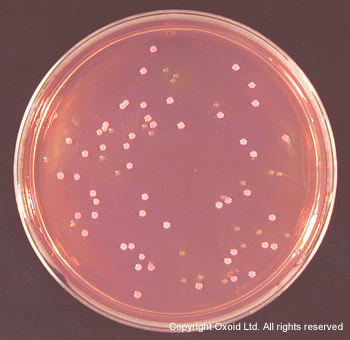
Featured Organism: Escherichia coli O157
SORBITOL MacCONKEY AGAR
Code: CM0813
a selective and differential medium for the detection of Escherichia coli O157
|
Typical Formula* |
gm/litre |
|
Peptone |
20.0 |
|
Sorbitol |
10.0 |
|
Bile salts No.3 |
1.5 |
|
Sodium chloride |
5.0 |
|
Neutral red |
0.03 |
|
Crystal violet |
0.001 |
|
Agar |
15.0 |
|
pH 7.1 ± 0.2 @ 25°C |
* Adjusted as required to meet performance standards
CEFIXIME-TELLURITE SUPPLEMENT
Code: SR0172
a freeze-dried supplement for use with Sorbitol MacConkey Agar for the selective isolation of Escherichia coli O157
|
Vial contents |
per vial |
per litre |
|
Potassium tellurite |
1.25mg |
2.5mg |
|
Cefixime |
0.025mg |
0.05mg |
Directions
Suspend 51.5g in 1 litre of distilled water. Bring to the boil to dissolve completely. Sterilise by autoclaving at 121°C for 15 minutes. Allow to cool to 50°C. Pour into sterile Petri dishes.
If required, reconstitute one vial of C-T supplement per 500ml medium, according to the instructions in the product leaflet. Aseptically add this to cooled medium before pouring into sterile Petri dishes
Description
Escherichia coli O157 is recognised as a cause of haemorrhagic colitis, an illness characterised by bloody diarrhoea and severe abdominal pain, and haemolytic uraemic syndrome (HUS)1, 2, 3, 4, 5, and as such, is a significant human pathogen.
Sorbitol MacConkey Agar is recommended for the isolation of pathogenic E. coli O157. The formulation, based on that described by Rappaport and Henig6, is identical to MacConkey Agar No.3, except that lactose has been replaced with sorbitol. E.coli O157 does not ferment sorbitol and, therefore, produces colourless colonies. In contrast, most E. coli strains ferment sorbitol and form pink colonies. The efficiency of Sorbitol MacConkey Agar has been confirmed by March and Ratnam7. These workers reported a sensitivity of 100% and a specificity of 85%, and recommended the medium as a simple, inexpensive, rapid and reliable means of screening for E. coli O157.
Chapman and co-workers8 added cefixime and potassium tellurite to Sorbitol MacConkey Agar to improve the selectivity of the medium. The level of potassium tellurite selects serogroup O157 from other E. coli serogroups and inhibits Providencia and Aeromonas species. Cefixime is inhibitory to Proteus spp.
The use of cefixime and tellurite in Sorbitol MacConkey Agar for isolation of E. coli O157:H7 is described in the FDA Bacteriological Analytical Manual 9.
Technique
1. Make up the agar according to the directions and pour into plates. If necessary dry the surface of the agar.
2. Inoculate the plates with a suspension of the test substance (food, faeces, etc) to produce separated colonies.
3. Incubate at 35°C for 24 hours. Doyle and Schoeni 10 reported that 35-37°C is the optimal temperature for growth of Escherichia coli O157. At 44-45.5°C growth is poor, even after 48 hours incubation.
Delay in reading plates beyond 24 hours should be avoided because the colour intensity of sorbitol-fermenting colonies fades, reducing the contrast with non-fermenting colonies. Other Gram-negative organisms including Pseudomonas, Proteus and Klebsiella species are able to grow on Sorbitol MacConkey Agar but may generally be differentiated by the appearance of their colonies.
A diagnostic reagent Escherichia coli O157 latex test (DR0620) is available so that instant confirmatory tests can be made from suspicious colonies.
Storage conditions and Shelf life
Dehydrated Sorbitol MaConkey Agar must be stored tightly capped in the original container at 10-30ºC.
Cefixime-Tellurite Supplement (SR0172) should be stored in the dark at temperatures below 0°C.
Oxoid Sorbitol MacConkey and Cefixime Tellurite Sorbitol MacConkey plates should be stored in the original packaging, at the temperature stated on the pack or product specification, and protected from direct light. When stored as directed, the unopened product will remain stable until the expiry date on the label.
Locally prepared media can be stored for up to 2 weeks when made from CM0813 and SR0172 according to the manufacturer’s instructions and stored at 2-8ºC, out of direct sunlight and protected from dessication. A longer shelf life may be attainable, but should be validated under the relevant, local manufacturing and storage conditions.
Appearance
Dehydrated medium: Straw/pink coloured, free-flowing powder
Prepared medium: Dark red gel
Quality control
Unsupplemented
|
Positive control: |
Expected results |
|
Escherichia coli O157:H7 Non-toxigenic NCTC12900 * |
Good growth; 1-2mm straw colonies |
|
Negative control: |
|
|
Escherichia coli ATCC® 25922 * |
Good growth; 1-2mm pink colonies |
Supplemented with CT Supplement SR0172 and tested in accordance with ISO 11133;201412.
|
Positive control: |
Expected results |
|
Escherichia coli O157:H7 Non-toxigenic NCTC12900 * |
Good growth; 1-2mm straw colonies |
|
Negative controls: |
|
|
Escherichia coli ATCC® 25922 * |
No growth/pinpoint-0.25mm pink colonies |
|
Escherichia coli ATCC® 8739* |
No growth/pinpoint-0.25mm pink colonies |
| Staphylococcus aureus ATCC®25923* WDCM00034 |
No growth |
| Staphylococcus aureus ATCC®6538* WDCM00034 |
No growth |
* This organism is available as a Culti-Loop®
Precautions
Although the great majority of Escherichia coli O157 strains have a typical appearance on Sorbitol MacConkey Agar, some strains are atypical 11. Sorbitol MacConkey Agar cannot be used solely to detect VTEC strains of Escherichia coli as some non-toxic strains will not ferment sorbitol 13.
Sorbitol MacConkey Agar and Cefixime Tellurite Sorbitol MacConkey Agar are in vitro diagnostic use only, by experienced microbiologists. They must not be used beyond the stated expiry date, or if the product shows any sign of deterioration.
Sterilise specimens, equipment and media properly after use.
Limitations
Micro-organisms with atypical enzyme patterns may give anomalous results.
References
1. Centers for Disease Control 1985 - United States, 1984, Morbid Mortal Weekly Rep., 34. 20-21.
2. Karmali M.A., Petric M., Lim C., Fleming P. C., Arbus G. S. and Lior H. (1985) J. Infect. Dis. 151. 775-782.
3. Karmali M.A., Steele B.T., Petric M. and Lim C. (1983) Lancet i: 619-620.
4. Pai C. H., Gordon R., Sims H. V. and Bryant L. E. (1984) Ann. Intern. Med. 101. 738-742.
5. Waters J. R. (1985) Can. Dis. Weekly Rep. 11. 123-124.
6. Rappaport F. and Henig E. (1952) J. Clin. Path. 5. 361.
7. March S. B. and Ratnam S. (1986) J. Clin. Microbiol. 23. 869-872.
8. Zadik P.M., Chapman P.A. and Siddons C.A. (1993) J. Med. Microbiol. 39. 155-158.
9. Food and Drug Administration (1995) Bacteriological Analytical Manual. 8th Edition. AOAC International. Gaitherburg. MD. Chapter 4, 20-23.
10. Doyle M. P. and Schoeni S. L. (1984) Appl. and Envir. Microbiol. 48. 855-856.
11.Karmali M. A. (1988) Culture 9. 2.
12.ISO 11133:2014 Microbiology of food, animal feed and water - Preparation, production, storage and performance testing of culture media
13.Lior H. and Borcryk A. (1987) Lancet. i. 333.
ATCC® is a registered trademark of American Type Culture Collection.
Copyright, Disclaimer and Privacy Policy | Conditions of Sale | About Us | Cookies
Thermo Fisher Scientific Inc.