Part of Thermo Fisher Scientific
Material Safety Data Sheet
Organisms
Organisms this product works with:
VET-RPLA TOXIN DETECTION KIT
Code: TD0920
A kit for the detection of Vibrio cholerae enterotoxin and Escherichia coli heat-labile enterotoxin in culture filtrates by reversed passive latex agglutination.
INTRODUCTION
Certain strains of Escherichia coli are known to produce enterotoxins. These enterotoxingenic Esch. coli (ETEC) strains are a common cause of diarrhoea in developing countries and of travellers’ diarrhoea. ETEC strains produce one or both of two different enterotoxins, a head-labile enterotoxin (LT) and a head-stable enterotoxin (ST). The LT enterotoxin as antigenic structures similar to those found on Vibrio cholerae enterotoxin (CT). Antiserum taken from rabbits, immunised with CT, will therefore react with both CT and LT1.
The VET-RPLA test is designed for the detection of LT or CT in culture fluid. A positive result given in the test indicates that the organism produces the relevant enterotoxin. The technique of reversed passive latex agglutination (RPLA) enables soluble antigen such as bacterial toxins to be detected in an agglutination assay.
In a standard agglutination assay, soluble antibody reacts with particulate antigen such as bacterial cells. However, in a reversed agglutination assay. the antibody, which is attached to particles, reacts with the soluble antigen. The particles (in this case, latex) do not, themselves, play a part in the reaction and they are therefore passive. The cross-linking of the latex particles by the specific antigen/antibody reaction results in the visible latex agglutination reaction.
PRINCIPLE OF ASSAY
Polystyrene latex particles are sensitised with purified antiserum taken from rabbits immunised with purified Vibrio cholerae enterotoxin. These latex particles will agglutinate in the presence of V. cholerae enterotoxin (CT) or Esc. coli heat-labile enterotoxin (LT). A control reagent is provided which consists of latex particles sensitised with non-immune rabbit globulins.
The test is performed in V-well microtitre plates. Dilutions of the culture filtrate are made in two rows of wells, a volume of the latex suspension is added to each well and the contents mixed. If either toxin is present, agglutination occurs which results in the formation of a lattice structure. Upon settling, this forms a diffuse layer on the base of the well. If the enterotoxins are absent or at a concentration below the assay detection level, no such lattice structure can be formed and, therefore, a tight button will be observed.
PRECAUTIONS
This product is for in vitro diagnostic use only.
Do not freeze.
Reagents with different lot numbers should not be interchanged.
Reagents and diluent contain 0.1% sodium azide as a preservative. Sodium azide may react with lead or copper plumbing to produce metal azides which are explosive by contact detonation. To prevent azide accumulation in plumbing, flush with copious amounts of water immediately after waste disposal.
STORAGE
The VET-RPLA Kit must be stored at 2-8° C. Under these conditions the reagents will retain their reactivity until the date shown on the kit box. After reconstitution the enterotoxin control should be stored at 2-8° C. Under these conditions, the reconstituted enterotoxin control will retain its reactivity for 3 months, or until the date shown on the kit box, whichever is the sooner.
SAMPLE PREPARATION
V.cholerae and Esch.coli for testing may be recovered from clinical samples and identified by suitable techniques described in standard textbooks.
It should be noted that Esch.coli is a normal inhabitant of the bowel and there may be more than one serotype present in the sample. It is therefore recommended that at least 6 colonies are separately tested to ensure a higher probability of detecting an enterotoxin-producing strain. Some ETEC strains produce LT in culture at a low level. Treatment of the broth culture with polymixin B should release sufficient LT for detection.
V.cholerae normally produces a large quantity of enterotoxin in culture and no special medium or extraction technique is require.
METHOD OF USE
Materials required but not provided
Microtitre plate (V-well) and lid
Fixed or variable pipette and tips (25ml)
Centrifuge capable of generating 900g (typically 3000rpm in a small bench top centrifuge) or membrane filtration unit using low protein-binding disposable filters with a porosity of 0.2mm - 0.45mm (such as Millipore SLGV)
Orbital Shaker
Culture media for promotion of enterotoxin production (suitable media are Peptone Water (Oxoid (CM9) adjusted to pH 8.4 for V.cholerae strains and Mundell’s Medium2.
Mundell’s Medium2 Formulation
Formula | gm/litre |
Casein Peptone (Oxoid L42) | 20.0 |
Sodium chloride | 2.5 |
Yeast Extract (Oxoid L21) | 6.0 |
Di-potassium hydrogen phosphate | 8.7 |
Dextrose | 2.5 |
Ferric chloride | 0.005 |
Manganese chloride | 0.005 |
Magnesium sulphate | 0.05 |
pH 8.5 ± 0.2 |
Polymixin B
Sodium hypochlorite solution (>1.3% w/w)
25ml dropper (optional)
25m l diluter (optional)
Micromixer (optional)
Moisture box (optional)
Components of the Kit
Instruction Leaflet
TD921 Sensitised Latex - Latex suspension sensitised with specific antibodies (rabbit lgG) against V.cholerae enterotoxin.
TD922 Latex control - Latex suspension sensitised with non-immune rabbit globulins.
TD923 Enterotoxin control - Dried V.cholerae enterotoxin.
TD924 Diluent - Phosphate buffered saline containing bovine serum albumin.
Toxin Production in Culture Fluid
Vibrio cholerae enterotoxin
A suitable medium is Peptone Water (Oxoid CM0009) adjusted to pH 8.4.
Inoculate the strain to be tested into the culture medium and incubate, preferably with shaking, at more than 110 rpm on an orbital shaker at 30°C for 24 hours.
After incubation, either centrifuge at 900g for 20 minutes at 4°C and use the supernatant as the test sample or membrane filter using 0.2µm - 0.45µm low protein-binding filter and use the filtrate as the test sample.
Escherichia coli enterotoxin
A suitable medium is Mundell’s medium 2.
Inoculate the strain to be tested into the culture medium (suitable volumes of medium would be 2 to 20ml) and incubate, preferably with shaking, at 37° C for 18 to 24 hours.
To the overnight broth culture (or a portion of it) add 3,4 polymixin B to a concentration of 10,000 units/ml. Incubate at 37°C for 4 hours. After incubation, either centrifuge at 900g for 20 minutes at 4°C and use the supernatant as the sample or membrane filter using a 0.2mm - 0.45mm low protein-binding filter and use the filtrate as the test sample.
Note: It is advisable to check the particular cultural method of use with standard toxin-producing strains of V.cholerae or Esch. coli such as Esch.coli NCTC11601.
Control
The reconstituted toxin control will agglutinate the sensitised latex. The use of the toxin control will provide a reference for the positive patterns illustrated below (see Interpretation of Test Results).
The control should be used from time to time only to confirm the correct working of the test latex. The toxin control is not provided at a specified level and therefore must not be used as a means of quantifying the level of toxin detected in the test sample.
Assay Method
Working Reagents
The latex reagents and diluent are ready for use. The latex reagents should be thoroughly shaken before use to ensure a homogeneous suspension. To reconstitute the enterotoxin control, add 0.5ml of diluent (TD924) to each vial. Shake gently until the contents are dissolved.
Arrange the plate so that each row consists of 8 wells. Each sample needs the use of 2 such rows.
Using a pipette or dropper, dispense 25ml of diluent in each well of the 2 rows except for the first well in each row.
Add 25ml of test sample to the first and second well of each row.
Using a pipette or diluter and starting at the second well of each row, pick up 25ml and perform doubling dilutions along each of the 2 rows. Stop at the 7th well to leave the last well containing diluent only.
Add 25ml of sensitised latex to each well of the first row.
Add 25ml of latex control to each well of the second row.
To mix the contents of each well, rotate the plate by micromixer or agitate by hand. Take care that no spillage occurs from the wells.
To avoid evaporation, either cover the plate with a lid or place the plate in a moisture box. Leave the plate undisturbed on a vibration-free surface at room temperature for 20 - 24 hours. It will help subsequent reading of the test if the plate is placed on black paper for the duration of the incubation.
Examine each well in each row for agglutination, against a black background.
Centrifuge tubes, membrane filters, microtitre plates, lids and pipette tips should be sterilised by autoclaving at 121° C or disinfected before disposal in hypochlorite solutions (>1.3% w/w)
Dispose of toxin controls and culture extracts in hypochlorite solutions (>1.3% w/w)
INTERPRETATION OF TEST RESULTS
The agglutination pattern should be judged by comparison with the following illustration:
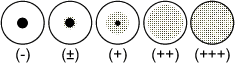
Results classified as (+), (++), and (+++) are considered to be positive.
Results in the row of wells containing latex control should be negative. In some cases, non-specific agglutination may be observed. In such cases the results should be interpreted as positive, provided that the reaction with sensitised latex is positive to a higher dilution of test sample than that seen with higher dilution of test sample than that seen with the control latex. The last well in all rows should be negative. If positive patterns are observed in some of these wells , the reaction should be regarded as invalid.
Note: With excess amount of CT, a prozone effect may be observed, i.e. negative pattern is obtained in wells containing test sample and sensitised latex. However, as a result of the doubling dilutions, the concentration of CT in each well along the row is progressively reduced and therefore the prozone effect due to excess amounts of CT is negated. A positive pattern of agglutination may, therefore, be seen after negative patterns in the first few wells of the row. With such results, the test sample should be classified as positive.
LIMITATIONS OF THE TEST
The sensitivity of this test kit in detecting CT is 1-2ng/ml. Enterotoxin present at concentrations lower than this will, therefore, give negative results.
REFERENCES
1. Ristaino, P.A., Levine, M., and Young. C.R. (1983). J. Clin. Microbiol. 18: 808-815.
2. Mundell, D.H., Anselmo, C.R. and Wishnow, R.M. (1976), Infection and Immunity 14: 383-388.
3. Evans, D.J. Evans, D.G and Gorbach, S.L. (1974), Infection and Immunity 8: 725.
4. Chapman, P. (1987.) Private communication.
FURTHER BACKGROUND INFORMATION
Almeida, R.J. Hickman-Brenner, F.W., Sowers, Evangeline, G., Puhr, Nance D., Farmer III J.J. and Wachsmuth, I.K. (1990) J Clin. Microbiol. 28: 128-130.
Copyright, Disclaimer and Privacy Policy | Conditions of Sale | About Us | Cookies
Thermo Fisher Scientific Inc.

