Part of Thermo Fisher Scientific
Material Safety Data Sheet
Organisms
Organisms this product works with:
For this Organism
Other products used in the isolation of Campylobacter:
Brilliance CampyCount AGAR
Code: PO1185
Brilliance CampyCount† Agar is a novel, selective and chromogenic medium for the enumeration of Campylobacter jejuni and Campylobacter coli from poultry and related samples.
| Typical Formula* | gm/litre |
| Defined salts mix | 18.9 |
| Amino acid mix | 3.7 |
| Agar | 12.0 |
| pH 7.6 ± 0.2 @ 25 ºC |
* Adjusted as required to meet performance standards
Description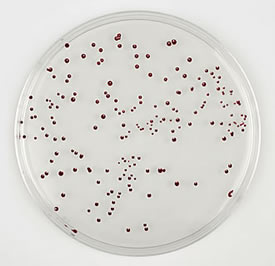 Campylobacter is a leading cause of enteric disease in most western countries and the worldwide commercial cost of the disease associated with poultry alone has been estimated at between €270M and €550M annually1. Poultry have been identified as a major causative agent in campylobacteriosis and the organism is endemic in many poultry populations, with Campylobacter jejuni and Campylobacter coli being responsible for the majority of infections.
Campylobacter is a leading cause of enteric disease in most western countries and the worldwide commercial cost of the disease associated with poultry alone has been estimated at between €270M and €550M annually1. Poultry have been identified as a major causative agent in campylobacteriosis and the organism is endemic in many poultry populations, with Campylobacter jejuni and Campylobacter coli being responsible for the majority of infections.
Although improvements in farming practices have reduced the prevalence of Campylobacter in poultry, it has not been completely eliminated, and as the source of flock infection is largely unknown, it appears that complete elimination of Campylobacter from poultry is unachievable in the short-term. Therefore, it is generally accepted that reducing the levels of Campylobacter on the bird is currently the best way to reduce the incidence of human infection, and international regulations will reflect this in the future.
Food testing for Campylobacter is predominantly conducted on a presence/absence basis, and uptake of enumeration methods has been limited by the lack of an appropriate medium, as well as vague, regulatory guidelines. The majority of Campylobacter infections are caused by consumption of under-cooked chicken, and as legislation moves towards enumerative limits on Campylobacter in poultry, food producers will put pressure on poultry companies to provide meat with low numbers of Campylobacter.
Brilliance CampyCount Agar is a new medium specifically designed for accurate, specific and easy enumeration of C. jejuni and C. coli from poultry testing. It is a transparent medium on which Campylobacter produce distinct dark red colonies, making identification and counting of Campylobacter significantly easier than on traditional charcoal or blood-containing agar. When used in conjunction with OBIS campy (ID0800) or Oxoid Campylobacter Test kit (DR0150), Brilliance CampyCount Agar can provide a confirmed Campylobacter count in as little as 72h.
 Technique
Technique
- Prepare samples in accordance with ISO 68872 or the appropriate standard
- Prepare required number of dilutions in a suitable diluent, such as Buffered Peptone Water ISO (CM1049) or Maximum Recovery Diluent (CM0733)
- In duplicate, spread 0.1ml aliquots of required dilutions over plates until absorbed
- Incubate the inverted dishes at 41.5°C ± 1°C for 48h ± 1h in a microaerobic atmosphere and then count any dark red colonies.
- Select five dark red colonies and subculture onto Columbia Blood Agar (CM0331)
- Incubate at 41.5°C for 24h in a microaerobic atmosphere
- Confirm by O.B.I.S. campy or methods such as morphology/motility, oxidase test, growth in air at 41.5°C and microaerobic growth at 25°C
- Report number of Campylobacter per gram of food.
For further instructions on the use and interpretation of Brilliance CampyCount Agar, simply download the Brilliance CampyCount Agar data sheet (1.7MB) in PDF format.
ISO 16140 Validation
Brilliance™ CampyCount agar has been validated and approved by MicroVal according to ISO 16140: 2003 standard against the reference method ISO/TS 10272-2: 2006 for the selective enumeration of thermotolerant Campylobacter species, in particular C. jejuni and C. coli, in poultry products. For flexibility this study included both the O.B.I.S. Campy kit and Oxoid Dryspot Campylobacter test as alternative confirmation methods to the confirmation methods as described in the reference method ISO/TS 10272-2: 2006.
MicroVal certificate no. MV2008LR12 is available in PDF format from www.microval.org.
Storage conditions and Shelf life
Brilliance CampyCount Agar plates should be stored in the original packaging at the temperature stated on the pack or product specification, and protected from direct light. When stored as directed, the unopened product will remain stable until the expiry date on the label.
Appearance
Prepared medium: transparent gel in Petri dishes
Quality Control
| Positive Controls | Expected results | % recovery* |
| Campylobacter jejuni ATCC® 33291 | 1-2mm, dark red colonies | 50% to 110% |
| Campylobacter coli ATCC® 43478 | 1-2mm, dark red colonies | 50% to 110% |
| Negative controls | ||
| Escherichia coli ATCC® 25922 | Minimal growth | <1% |
| Staphylococcus aureus ATCC® 25923 | Minimal growth | <1% |
| Candida albicans ATCC® 10231 | Minimal growth | <1% |
*Compared to Columbia Blood Agar
Precautions
Brilliance CampyCount Agar is especially light sensitive. Protect from direct sunlight as much as possible. Ensure plates are dried thoroughly before use to prevent spreading of Campylobacter colonies.
Limitations
Species of Acinetobacter may grow as dark red colonies on the plate. These are easily distinguished as they appear far larger than Campylobacter (usually >3mm in diameter) and are surrounded by a clear mucoid halo. The MicroVal study revealed that in chicken thighs Brilliance™ CampyCount agar plates gave a lower yield than mCCDA plates.
References
1. WHO, Report and Proceedings of WHO consultation of experts. 2001
2. ISO 6887-1:1999 Microbiology of food and animal feeding stuffs - Preparation of test samples, initial suspension and decimal dilutions for microbiological examination
ATCC® is a registered trademark of American Type Culture Collection
Copyright, Disclaimer and Privacy Policy | Conditions of Sale | About Us | Cookies
Thermo Fisher Scientific Inc.

